SCOPE2 PET scan study
15 June 2023
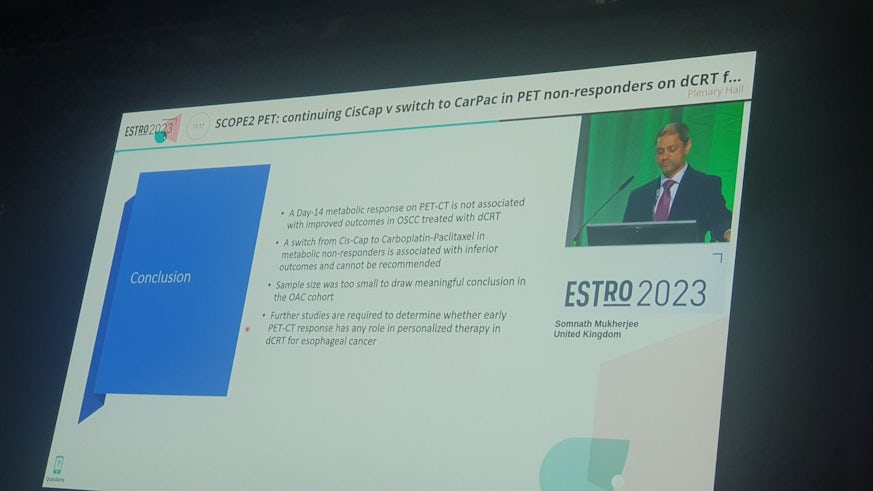
SCOPE2 is a UK cancer clinical trial for patients with locally advanced oesophageal cancer (cancer of the food pipe). The trial explores the optimum dose of radiotherapy to give to patients to optimise their chance of recovery. SCOPE2 includes a sub-study which is now complete, the results of which were presented at an international cancer conference in Vienna on the 13th May 2023. The aim of this smaller sub-study was to see whether using a specialist scan called a PET (Positron Emission Tomography) scan to assess early changes in a patient’s cancer whilst he/she is receiving chemotherapy can guide a more personalised treatment approach, thereby increasing the number of patients who are cured. All patients were checked at 24 weeks after the start of treatment to see how they had responded.
Doctors usually give patients with locally advanced oesophageal cancer two drugs called cisplatin and capecitabine for a few weeks prior to starting radiotherapy. Since this standard treatment doesn’t work for all patients, the trial wanted to test whether changing the drugs to carboplatin and paclitaxel would improve outcomes for these patients.
Participants in this sub-study had a PET scan 14 days after they started chemotherapy, as data from other research showed that this identified patients who would not respond well to treatment. This scan would then be compared with the original PET scan that the patient had when their cancer was first diagnosed to see whether the patient was responding to the first two weeks of their chemotherapy treatment. If there was little or no response they were randomised to either continue receiving the same chemotherapy regimen (which would be the current standard), or to switch to the new combination of drugs for the rest of their treatment. As well as receiving either their original or switched chemotherapy, they would also receive either a standard dose or higher dose of radiotherapy alongside the last 2 cycles of their chemotherapy. A total of 103 patients were recruited into this sub-study, 63 of whom did not respond well to their first 14 days of chemotherapy.
The sub-study has now been analysed and the results were recently presented at the leading European radiotherapy conference called ESTRO (#ESTRO2023). The study shows that a lack of a response on a PET scan 14 days into treatment does not identify a group of patients who do worse with chemotherapy and radiotherapy to treat their locally advanced oesophageal cancer. The results also suggest that it is best not to switch the chemotherapy treatment in the group of patients who do not show a response on the PET scan, as switching the chemotherapy regimen based on the response on the PET scan at 14 days may actually be slightly worse for patients. This switch is therefore not recommended.
Prof Richard Adams, Director of the Centre For Trials Research, says “the results from the SCOPE2 sub-study presented in Vienna demonstrate the importance of undertaking rigorous assessment of new approaches to treatment before changes are made. Our research shows that, currently, doing a Day 14 PET-CT to assess for response is not helpful and therefore not recommended. The SCOPE2 trial continues to recruit patients to explore the main question of whether an increase in radiotherapy dose can improve survival in patients with locally advanced oesophageal cancer. We are indebted to patients and their families for their ongoing support in helping us improve the care of patients in the future”.
SCOPE2 is funded by Cancer Research UK and sponsored by Velindre University NHS Trust. The trial is run by the Centre for Trials Research (Cardiff University) with core funding provided by Health Care Research Wales and Cancer Research UK. The Chief Investigator is Professor Tom Crosby, and Prof Somnath Mukherjee is the lead for the PET sub-study. We would like to express our deep gratitude to all participants for their involvement in this research.
For further information please contact SCOPE2@cardiff.ac.uk.